About Sight Glass

On the left is fused sight glass with partially filled metal housing. On the right is full height sight glass, where the glass is flat on both sides of the fitting. Notice optical clarity and no viewing distortion.
Sight glass credit: Metaglas®. Photo credit: Encole LLC.
The objective of a sight glass is to provide a window into a pressure vessel, a hot temperature or corrosive process environment, for the purposes of machine vision or live observation.
Sight glass for process observation in industrial applications is a term of art. The materials for the glass and the metal are chosen based on corrosion requirements, operating temperature, pressure, and the desired wavelength to be transmitted through the glass. Typical glass materials used in sight glasses are sodalime for low cost applications, borosilicate for higher temperatures, or crystalline materials for wide transmission of wavelengths and superior mechanical properties. Typical metal used in sight glass is a type of stainless steel chosen for required strength, and compatibility with corrosion requirements, or a metal alloy where glass to metal thermal expansion difference is taken into consideration, typically for larger size sight windows.
Sight Glass Materials Compatibility
| Housing Material | Window Material | Glass Sealing Method | Max Temp. | Max Pressure, psi* | Corrosion Resistance | Transmitted Wavelength** | ||
|---|---|---|---|---|---|---|---|---|
| Brine, NaCl | H2S gas | Fluorine | ||||||
| Stainless 303 | Soda Lime | Fused | 160 C | > 40000 | Poor | Poor | Poor | 350nm - 2500nm |
| Stainless 304 | Borosilicate | Fused | 300 C | > 40000 | Poor | Poor | Good | 350nm - 2500nm |
| Stainless 316 | Borosilicate | Fused | 300 C | > 40000 | Good | Good | Poor | 350nm - 2500nm |
| Stainless 17-4 | Borosilicate | Fused | 300 C | > 40000 | Poor | Good | Poor | 350nm - 2500nm |
| Stainless 440 | Borosilicate | Fused | 300 C | > 40000 | Poor | Poor | Poor | 350nm - 2500nm |
| Duplex Steel 2205 | Borosilicate | Fused | 300 C | > 40000 | Excellent | Excellent | Good | 350nm - 2500nm |
| Hastelloy C-22 | Borosilicate | Fused | 300 C | > 40000 | Excellent | Excellent | Good | 350nm - 2500nm |
| Hastelloy C-276 | Borosilicate | Fused | 300 C | > 40000 | Excellent | Good | Good | 350nm - 2500nm |
| Hastelloy C-276 | Fused Quarts | Organic Sealing | 250 C | > 40000 | Excellent | Good | Good | 180nm - 3500nm |
| Titanium Beta-C | Fused Quarts | Organic Sealing | 250 C | > 40000 | Excellent | Excellent | Excellent | 180nm - 3500nm |
| Hastelloy C-276 | Fused Silica | Organic Sealing | 250 C | > 40000 | Good | Good | Good | 160nm - 3500nm |
| Stainless 316 | Sapphire | Organic Sealing | 250 C | > 40000 | Good | Good | Excellent | 170nm - 6500nm |
| Hastelloy C-276 | Sapphire | Organic Sealing | 250 C | > 40000 | Good | Good | Good | 170nm - 6500nm |
| Titanium Beta-C | Sapphire | Organic Sealing | 250 C | > 40000 | Excellent | Excellent | Good | 170nm - 6500nm |
| Duplex Steel 2205 | Zinc Selenide | Organic Sealing | 150 C | 5000 | Excellent | Excellent | Good | 0.12µm - 21µm |
| Stainless 304 | Calcium Fluoride | Organic Sealing | 150 C | 5000 | Poor | Poor | Good | 150nm - 9000 nm |
| Stainless 304 | Magnesium Fluoride | Organic Sealing | 250 C | 5000 | Poor | Poor | Good | 120nm - 9000 nm |
| Niobium | Sapphire | High Temp Brazing | 800 C | > 30000 | Excellent | Excellent | Good | 170nm - 6500nm |
| Kovar | Borosilicate | Fused | 400 C | > 40000 | Excellent | Excellent | Good | 350nm - 2500nm |
| Invar | Fused Quarts | Organic Sealing | 150 C | > 40000 | Excellent | Excellent | Good | 180nm - 3500nm |
| Invar | Fused Silica | Organic Sealing | 150 C | > 40000 | Excellent | Excellent | Good | 160nm - 3500nm |
* - Maximum pressure depends on housing design and window thickness.
** - Transmittance % depends on thickness of window material.
Glass Sealing Methods

Glass is flush with the top surface of the metal fitting. Sight Glass credit: L.J. Star Inc. Image credit: Encole LLC.

Notice the glass is flat on both sides, top and bottom.
Fused Glass Sealing
Standard or specialty glass is thermally fused directly to a metal housing. Typically sealed into NPT and BSPT housings, these sight window / sight glass systems can be as strong as the equivalent size solid metal NPT or BSPT plug (strength determined by glass thickness). Windows can also be fused into other types of housings, such as: flanges and tubes. These sight windows/sight glasses have been tested and operated at temperatures from cryogenic to in excess of 400 C and pressures up to 40,000 PSI.
Common applications - level gauges, sight indicators, flow gauges in refrigeration and hydraulics systems, furnace and heater systems, pilot and fire windows.
Usage - where high resolution optical clarity is not required, but reliability and low cost are.
Organic Sealing
Glass, sapphire and other crystalline windows are sealed directly into metal or ceramic housings via proprietary organic sealing systems. In some applications, organic sealing is superior to metal brazing if operating temperatures do not exceed 250 C. The organic sealants are NASA approved for zero out-gassing and are also highly chemical resistant.
Common applications - High pressure research, medial applications, optical analysis, particle counting and measurement, laser windows, plasma chambers, cryo analysis, petroleum research and downhole applications.
Usage - Lower cost than brazing, where high resolution optics are required along with high strength and reliability. Also good for CTE mismatches where flexible sealing systems are required.
Braze Sealing
Glass, sapphire and other crystalline windows are brazed directly into metal or ceramic housings. The process is similar to organic sealing except, in lieu of a sealant, high temperature brazes are used. Typical housings are NPT and BSPT fittings, but windows can also be brazed into tubes, flanges and customer designed housings. Depending on design, these windows can be subjected to over 1100 C and 40,000 PSI.
Common applications - Pyrometry, high pressure research, optical analysis, supercritical applications, medical applications, particle counting and measurement, laser windows, plasma chambers.
Usage - where high resolution optics are required along with high strength and reliability.
Compound Annular Sealing
Specialty engineered compound annular sight windows / sight glasses are optimal for high pressure and hazardous applications. Used when evaluation and observation must be performed by camera or fiber optics viewing and proper lighting is critical. Made from sapphire, these windows are highly durable, scratch and chemical resistant. These superior viewing windows have an outer ring for illumination and an inner optical window for viewing. Pressure range upward of 20,000 PSI.
Common applications - down-hole camera systems, deep sea robotics, petroleum platform inspection, pipe inspection, hazardous inspection systems.
Usage - for high resolution viewing in hostile environments where proper lighting is critical.
Glass and Crystalline Material Differences
What is the difference between glass and crystalline material? Glass is an amorphous solid which means its atoms are randomly oriented. Whereas, a crystalline material may contain the exact same type and quantity of atoms as glass, but the atoms are ordered in rigid, well-defined patterns. Fused quartz glass vs. crystalline quartz is a good example of the same type of atoms (SiO2) structured in such a way that one orientation forms a glass and another forms a crystal.
Crystalline materials are solid and keep their shape until they reach a very specific temperature, at which time they become fluid. In contrast, all glasses, by definition, are always a fluid. The viscosity of glass is determined by the temperature of the glass; even at room temperature glass is moving, albeit very slowly. When product tolerances are very tight and temperatures are elevated, a crystalline material is superior to glass because it will maintain its shape at any temperature below the melting point. Conversely, glass will change shape over time, regardless of the melting point and especially at elevated temperatures.
Both crystalline solids and glasses are normally colorless in the pure state. Impurities are actually what give sapphire and glass their color. The impurities also change the mechanical, thermal, and optical properties of both material types, especially for glass.
Glass Materials Relative Properties
| Property, Characteristic | Sapphire | Fused Quartz | Fused Silica | Borosilicate Glass | Soda Lime Glass |
|---|---|---|---|---|---|
| Solarization | No | Very low | Very low | Yes | Yes, the most |
| Sags of slumps at temperature | No | Somewhat | Somewhat | Yes | Yes, the most |
| Corrosion-resistant in most acids | Yes | Somewhat | Somewhat | Somewhat | No |
| Corrosion-resistant in fluorinated acids | Yes | Somewhat | Somewhat | No | No |
| Corrosion-resistant in plasmas | Yes | Somewhat | Somewhat | Somewhat | No |
| Can be bent, molded or shaped | No | Yes | Yes | Yes | Yes |
| Devitraification when molding | - | No | No | No | Yes |
| Can be tempered | No | No | No | No | Yes |
| Cost | $$$$ | $$$ | $$$ | $$ | $ |
| Size limitations | Limited | No | No | No | No |
Sapphire

Sensor head of reflection scan process spectometer for analyzing liquids, powders or bulk materials. Image credit: Polytec GmbH. Sapphire window credit: Encole LLC.

Quartz Sight Glass with Organic Seal Under 6400 psi Hydrostatic Pressure. Image credit: Encole.

Oil level observed with NPT 2-inch sight glass, Quartz Window. Image credit: Stanford Linear Accelerator Center, SLAC.

Thermal and pressure testing of a sapphire sight glass using a metal seal. Metal seal used is a C-seal type for internal pressure, credit: Jetseal, Inc. Image credit: Encole.
Sapphire is a single crystal - Aluminum Oxide (Al2O3) - which is colorless and optically clear. Synthetic sapphire is grown in boules (bulk pieces) no larger than 300mm in diameter. It can also be grown into shapes such as sheets, ribbons, domes and tubes with very smooth surface qualities, high purity and optical translucence. When used "as grown" there is very little need for grinding and polishing.
Why Sapphire?
- Mechanically second only to diamond. One of the hardest and most scratch resistant materials available. The high modulus of elasticity and high tensile strength make it extremely wear, abrasion and impact resistant.
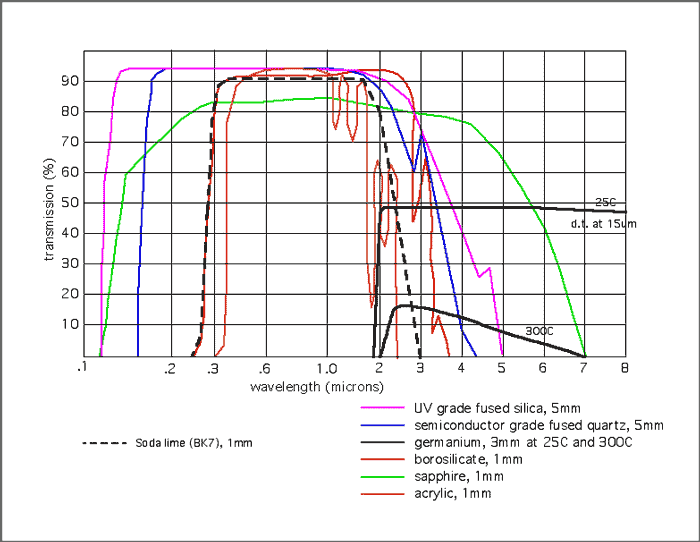
- Colorless optical characteristics are superior to any standard glass, with up to a 98.5% transmission and a transmission window from 190 nanometers in the UV to 5 microns in the IR.
- No solarization in high-radiation systems.
- High dielectric constant and low loss tangent makes it a great electrical insulator and low loss long wavelength window.
- Thermally very stable. Does not lose any of its mechanical and optical qualities from cryogenic to over 2000 C.
- Thermal conductivity greater than other optical materials and most dielectrics.
- No surface damage and devitrification due to extreme thermal cycling.
- Does not sag or slump at very elevated temperatures.
- Highly corrosion resistant. More resistant to corrosive chemicals than most standard hard materials available.
Why Not Sapphire?
- Cost of material. Sapphire often costs more than other refractory materials. But not always! Quantity and geometry play a major role in the cost of the final product, especially with smaller products where the labor is the primary cost.
- Cannot be bent, molded, drawn or melt-fused like glasses and metals. Sapphire can only be ground and mechanically polished.
- Size limitations. The maximum size of a sapphire product cannot exceed the largest boule that can be grown. Therefore, maximum part size cannot exceed 300mm for two of the dimensions.
- Larger pieces can be thermally shocked and broken if not heated uniformly.
Fused Quartz and Fused Silica Glass
Fused quartz and fused silica are the amorphous form of quartz. Fused quartz is made from purifying and melting natural crystalline quartz, usually natural quartz sand. Fused silica is a purer version of fused quartz that is made from various silicon gasses. Chemically known as SiO2, silica is "pure" glass. All other commercial glasses are SiO2 with other dopants added to lower the melting temperature and modify optical, thermal and mechanical characteristics.
Why Fused Quartz and Fused Silica Glass?
- Extremely low coefficient of expansion, making it far more shock resistant than any other refractory material.
- Best transmission characteristics of any standard glass: 220 nanometers to 3 microns for standard semiconductor-grade fused quartz, and 175 nanometers to 3 microns for many types of fused silica.
- Highest temperature characteristics of any glass. A continuous maximum of 900 C to 1100 C, depending on the size and shape of the part. Can be used up to 1400 C for short periods of time.
- Very low dielectric constant and the lowest loss tangent of almost all known materials.
- Very low thermal conductivity.
- Can be melted, bent, fused, drawn and welded into tube and rod forms.
- Can be ground and polished.
- With the right technology, can be molded, slumped and drawn into fiber, tube and rod shapes.
- Harder than most glasses.
- Can be made into any shape and relatively large sizes.
- Excellent resistance to non-fluorinated acids, solvents and plasmas.
- Excellent for containing many high-purity chemicals.
- Less expensive than sapphire for larger parts.
Why Not Fused Quartz and Fused Silica Glass?
- Much more costly than other standard glasses.
- Can sag and slump over time at elevated temperatures.
- Surface devitrifies over time when temperature is cycled at high temperatures.
- Breaks down with some caustics, fluorinated acids and plasmas.
- Can solarize in high radiation environments.
- Due to the high melting point, fabrication costs for melting and blowing are much higher than other standard glasses.
- Standard shapes are tubes and boules. Does not come in standard sheets like borosilicate and soda lime glasses. In other words, other than tubes, all fused quartz and silica products must be ground and polished from a large block.
Borosolicate Glass
Borosilicate glass is an "engineered" glass developed specifically for use in laboratories and applications where thermal, mechanical and chemical conditions are too harsh for standard, household-type glass. Some common names of borosilicate are Pyrex™ by Corning and Duran™ or borofloat™ by Schott Glass. Like most glasses, the main component of borosilicate glass is SiO2 with boron and various other elements added to give it its excellent qualities.
Why Borosilicate Glass?
- Much easier to hot-work than quartz, making fabrication less costly.
- Material cost is considerably less than fused quartz.
- Compared to all glasses, except fused quartz, it has a low coefficient of expansion (three times less than soda lime glass). Useful for cooking, heating and other thermal environments, without the risk of breakage due to thermal shock.
- Like soda lime glass, the float process is used to make relatively low-cost, optical-quality sheet borosilicate glass in a variety of thicknesses (less than 1mm to over 25mm thick).
- Easily moldable (compared to quartz).
- Minimum devitrification when molding and flame working. High-quality surfaces can be maintained when molding and slumping.
- Thermally stable up to 450 C for continuous use, and up to 600 C for short periods.
- More resistant to non-fluorinated chemicals than household soda lime glass.
- Mechanically stronger and harder than soda lime glass.
Why Not Borosilicate Glass?
- Material will not maintain its shape if exposed to temperatures greater than 450 C for long periods of time.
- Cost. Borosilicate is usually 2 to 3 times more expensive than soda lime material.
- Not as thermal shock resistant as quartz, which has a coefficient of expansion ~60x less.
- For high purity chemicals, some minor leaching can occur over time, especially if exposed to some acidic or basic chemicals.
- Cannot be fully tempered like soda lime glass.
Soda Lime Glass
Soda lime glass is the "original" glass, appearing in its most basic form thousands of years ago. Commonly called float glass, it is often formed by floating soda lime glass on a bed of molten tin. It is also knows as crown glass, a high-silica form of soda lime that was historically used for windows. Soda lime glass is composed of SiO2: sodium oxide (soda) and calcium oxide (lime). About 90% of the glass used in the world - including most windows, dinnerware, art and lighting products - is one of 50,000 variations of soda lime glass.
Although soda lime glass typically has a green or blue-green tint to it, the iron content can be reduced to the point where the glass becomes crystal clear, also known as "water white."
Why Soda Lime Glass?
- Inexpensive and easy to mass produce.
- Low melting temperature; maintains softness for a long time. This allows for long working times and faster production rates.
- Easily "floated," making it a very low-cost, flat (float), optically clear sheet glass.
- Softer than borosilicate and quartz, making scribe cutting easier and faster.
- Because of its high coefficient of expansion, it is easily tempered. Tempered glass is up to 3 times stronger than non-tempered glass and it crumbles when broken - a good (and often required) safety feature.
Why Not Soda Lime Glass?
- High coefficient of expansion, thus, very poor thermal shock resistance. Only good in thermal environments where heating is uniform and gradual.
- Will sag easily at relatively low temperatures.
- Is not available in as many stock thicknesses as borosilicate.
- Many chemicals will leach the glass over time, making it unsuitable for pure chemical applications.
- Not as scratch resistant as borosilicate and quartz.
Glass Ceramic
Glass ceramic is a clear, light amber-tinted or opaque black material. It comes stock in only 3mm, 4mm and 5mm thicknesses. It has many of the thermal performance properties of fused quartz including a very low coefficient of expansion. For fabrication capabilities, it is more like sapphire, as it cannot be melted or welded. It is not nearly as good for optical applications as fused quartz, sapphire, soda lime or borosilicate. However, it is ideal for cooktops, furnaces and fireplaces with windows where the tint of the glass ceramic is not a cosmetic problem.
Transmission Curves
Sight Glass Pressure Ratings
Sight windows / sight glasses can be manufactured from ultra-high vacuum to pressures in excess of 40,000 PSI. Pressure ranges are determined by: (1) glass sealing method into the housing, (2) housing design, (3) housing material, (4) glass material and thickness.
